 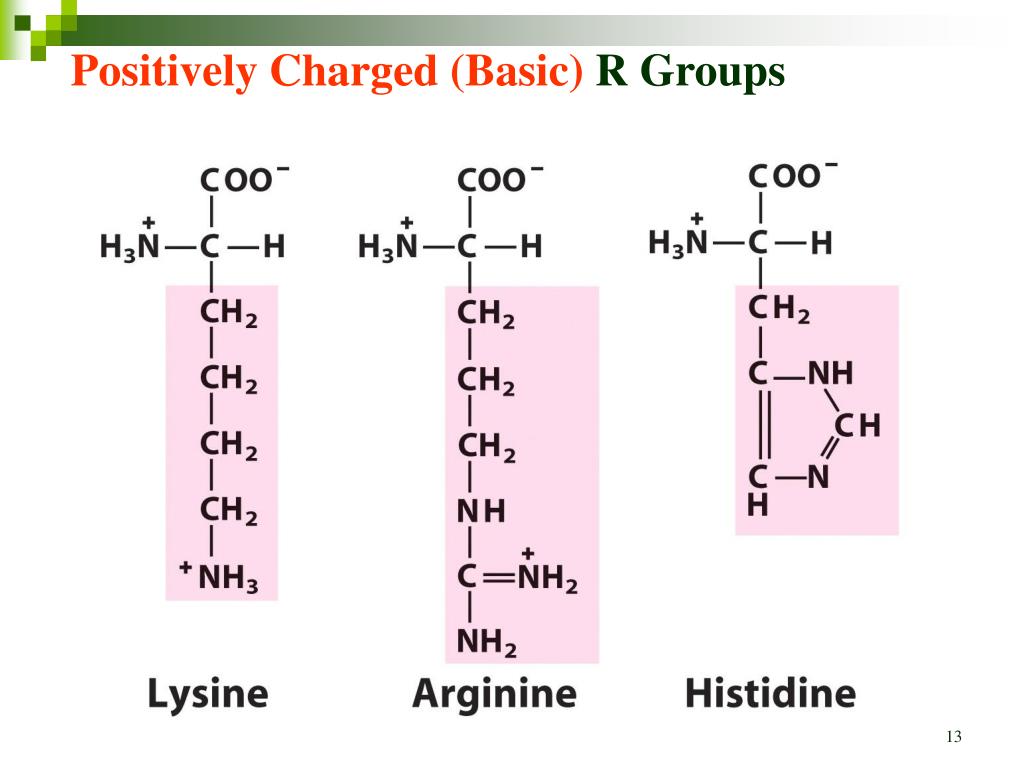
The pKa values for the ionizable groups are shown on the graph, with each group represented by a different color (Lys=green, Gln=purple, Trp=orange). The pK values for R-groups vary with the ionic environment. The pK and pI values of selected amino acids are listed in Table 3.2. The x-axis represents pH, and the y-axis represents molarity (mol/L). The only amino acids with R-groups that have buffering capacity in the physiological pH range are histidine (imidazole pK6.0) and cysteine (sulfhydryl pK8.3). The titration curve for this peptide is shown in Figure 1. Draw the appropriate titration curve for this peptide on graph paper starting at pH 0 and ending at pH 14. Therefore, the pKa values for the ionizable groups are 4.83, 7.41, and 2.68, respectively.ģ. The pKa values for the Lys, Gln, and Trp residues are 5.76, 8.17, and 5.76, respectively. How many ionizable groups does his peptide have? Therefore, the peptide has three ionizable groups (Lys-Glu-Trp).Ģ. The pKa value for the Trp residue is 5.76. The pKa values for the Lys and Gln residues are 8.17 and 10.93, respectively. The Trp residue is in turn bonded to the Glu and Phe residues. This function calculates the amount of amino acids of a particular class and classified as: Tiny, Small, Aliphatic, Aromatic, Non-polar, Polar, Charged, Basic and Acidic based on their size and R-groups using same function implemented in EMBOSS pepstat. Isoelectric point definition: Isoelectric point (pI) is a pH in which net charge of protein is zero. In the structure, the Lys and Gln residues are each bonded to a Trp residue. Draw the peptide structure when all the ionizable groups are fully protonated. (Watch on YouTube: Isoelectric Point of Amino Acids. This is usually a trap answer on tests.1. This video shows you how to calculate amino acid isoelectric point along with a shortcut to help you figure out WHICH pKa to use when given a side chain pKa value. Note that this is the WRONG structure at a neutral pH. Polar but uncharged R groups Serine Threonine Cysteine 4. Aromatic, R groups Leucine Tyrosine Isoleucine Tryptophan Methionine Phenylalanine 3. 1.Non polar, aliphatic R groups: Glycine Alanine Proline Valine 2. Here is an example of deprotonating a protonated R group (glutamic acid). Before that we classify the amino acids on the basis of their R group. 3 pKa - 3 ionisable groups How to calculate the pl when the side chain is ionisable o Identify species that carries a net 0 charge o For histidine take the average of the 2 pka values of the amino and imidazole groups (+vely charged groups when protonated): I. If you start at a high pH and decrease it, read the structures from right to left. Here is the structures at increasing pHs for a generic amino acid. The #pK_b# values for amino groups are lower than that of carboxyl groups, so the amino groups will be protonated before the carboxyl groups. The exact opposite would happen for protonation of amino acids. Use the following link to find a list of the #pK_a# values for all the amino acids. The atom with the lowest #pK_a# will be deprotonated. According to orthocresols answer: Since the p I is the p H at which the amino acid has no overall net charge, you need to average the p K a values relevant to the protonation/deprotonation of the form with no net charge. The calculation is based on the pK values of the amino acids and of the terminal amine & carboxyl group of the peptide chain. At this pH the the amino acid sequence forms a zwitterion. As pH increases, it will be deprotonated before the ammonium group.įor the amino acids with protonated R groups, you need to pay attention to their #pK_a# values. The isoelectric point is defined as the pH at which a particular amino acid sequence (peptide chain) bears no net electrical charge. The #pK_a# of the carboxylic acid is always lower than that of the ammonium group. Look at the #pK_a# values of the ammonium and carboxyl groups. This is due to ammonium (amino) groups being less acidic than carboxylic acids. Amphoteric, dipolar species are called zwitterions. Amino acids are amphoteric, meaning they can act like an acid and base. 
ago Technically it's the average of the two pKa's that surround the neutral zwitterion. Then just figure out which combination gives a neutral amino acid, and the PI can be determined from that. Notice something weird? The amino group is protonated but the carboxyl is not. At a pH that's equal to a given pKa, the group can be protonated with decreasing pH or deprotonated with increasing pH. Let's start by looking at the generic structure of an amino acid. The pH of an amino acid affects which atoms protonate and deprotonate. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
